In case of ammonia, when we dissolve ammonia in any aqueous solution like water ammonia does not dissociate in it completely and also produce less OH- ions. As the condition for weak base is says that when the molecule is incompletely or sparingly or partially soluble in aqueous solution and does not produce complete OH- ions then the molecule behaves as a weak base. 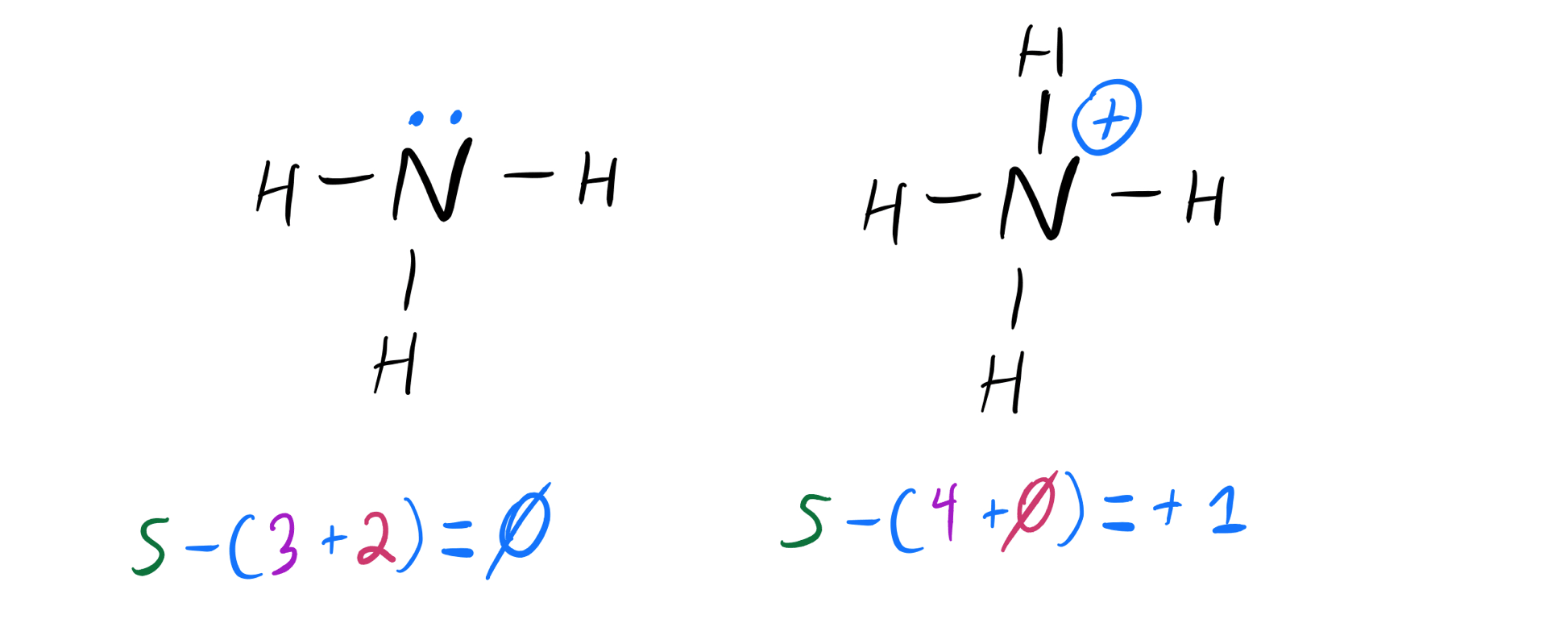
Thus, NH3 (ammonia) is not a strong base as the ammonia molecule are not dissociated completely in aqueous solution and also it produce very less amount of OH- ions. not all molecules get dissociates in a solution to produce OH- ions are weak base and also at equilibrium there is both ionised product and dissociated molecules are present in the solution. 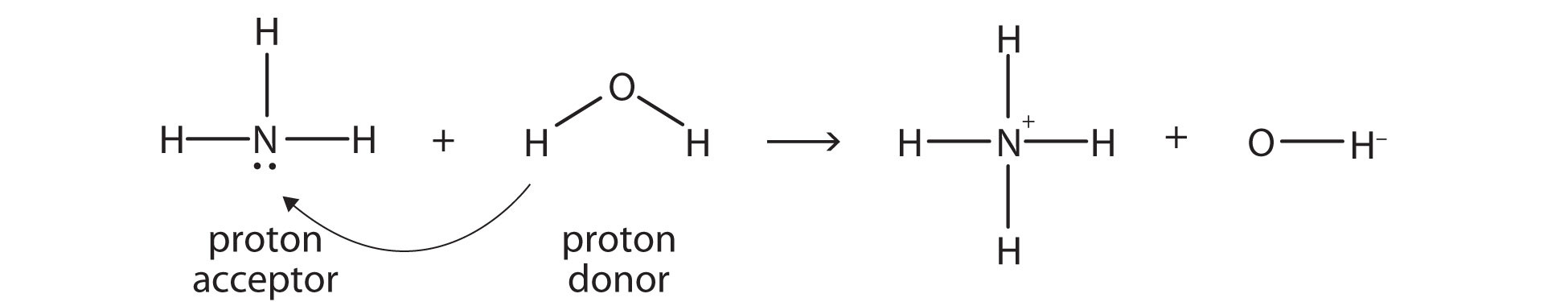
The compounds when incompletely or partially disassociates into aqueous solution i.e. Ex: Potassium hydroxide (KOH), sodium hydroxide (NaOH). All strong base molecules get disassociates into OH- ions and nothing remain in solution for dissociation. To check whether NH3 is a strong base or not, let’s take a look on the definitions of strong base and weak base.The compound when fully disassociates in aqueous solvents or solutions, it produce hydroxide (OH-) ions. Is NH3 a strong base?Īmmonia is not a strong base. So as per acid base lewis theory, NH3 molecule is considered as a lewis base because it has lone electron pairs on it. Also it donates H+ ions to forms NH2- ions which are a conjugate base and NH3 behaves as a acid. NH3 by taking H+ ion behaves as conjugate acid by forming NH4+ ions which shows it is as a weak acid. When we added ammonia in water it takes H+ ions from water and donates its lone electron pair to form OH- ions due to which it shows basic nature. But NH3 is also amphoteric in nature which shows both acid and base character in specific conditions. It has pH value 11 shows its basic nature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
